A) 75.4 h
B) 68.4 h
C) 74.0 h
D) 12.4 h
E) 18.7 h
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the radioactive nuclide 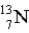 undergoes positron emission,what is the product nuclide?
undergoes positron emission,what is the product nuclide?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
E) ![]()
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The symbol Ga stands for the element
A) gallium
B) germanium
C) gold
D) gadolinium
E) none of these
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following terms was not discussed by Dalton in his atomic theory?
A) Only isotopes
B) Ions and protons
C) Protons,electrons,and neutrons
D) Only neutrons
E) Isotopes,ions,protons,electrons,and neutrons
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many electrons are present in a tin atom with a mass number of 119?
A) 50
B) 119
C) 169
D) 69
E) 6.02 × 1023
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following does Dalton's Atomic Theory not address?
A) Hydrogen and oxygen gas combine to form water.
B) H2O2 consists of 16 g of oxygen for every 1 g of hydrogen.
C) The seat you are sitting on is made up of atoms.
D) Hydrogen ignites in an open flame but helium does not.
E) The relative mass of a carbon atom is about 12 times that of hydrogen.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which particle does the nuclide symbol 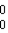 b represent?
b represent?
A) electron
B) positron
C) gamma ray
D) proton
E) helium nucleus
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a metalloid?
A) Antimony
B) Gallium
C) Silicon
D) Germanium
E) Arsenic
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Determine the number of protons and the number of neutrons in an atom of Sn-118.
Correct Answer

verified
50 protons...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
How many neutrons are contained in an iodine nucleus with a mass number of 130?
A) 53
B) 73
C) 77
D) 126
E) 130
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The atom with 69 neutrons and 50 protons has a mass number of
A) 69
B) 50
C) 19
D) 119
E) cannot be determined from information given
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which atomic particle determines the chemical behavior of an atom?
A) proton
B) electron
C) neutron
D) nucleus
E) none of these
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The symbol for the element calcium is
A) Ca
B) Co
C) Cu
D) C
E) Cl
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom with 15 protons and 16 neutrons is an atom of
A) P
B) Ga
C) S
D) Pd
E) Rh
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the most abundant metal on the earth (including the crust,oceans,and atmosphere) ?
A) silicon
B) oxygen
C) aluminum
D) carbon
E) iron
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following sets of nuclei is the most commonly used fissionable isotopes in nuclear reactors?
A) ![]() and
and ![]()
B) ![]() and
and ![]()
C) ![]() and
and ![]()
D) ![]() and
and ![]()
E) ![]() and
and ![]()
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
Technetium-99m is routinely used in medical imaging.The italics m means the nucleus is ________.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a nonmetal?
A) cerium
B) cesium
C) carbon
D) calcium
E) copper
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many atoms of hydrogen are in three molecules of C6H13Cl?
A) 39
B) 3 x 1023
C) 13
D) 6 x 1023
E) 13 x 1023
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
By what (single step) process does americium-241 decay to neptunium-237?
A) α particle emission
B) β particle emission
C) Positron emission
D) Electron capture
E) Neutron capture
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 190
Related Exams